Introduction and comparison of battery grade lithium hydroxide
Aug,06,24

Lithium has a complete industrial chain, forming lithium carbonate, lithium hydroxide, and mid stream lithium metal from ores and salt lakes in the upstream.
Moving on to downstream traditional industries such as metal smelting, lubricants, ceramics,
and glass New materials (organic synthesis, biomedical) and new energy (3c batteries, batteries, etc.).,
For use by large 3C lithium battery manufacturers.
This may refer to the top 10 3C consumer lithium battery manufacturers And a complete industry chain in other application areas.
The production process of lithium hydroxide, a lithium battery material, was introduced, and the analysis of lithium carbonate was compared.
Introduce battery grade lithium hydroxide
Lithium hydroxide is one of the three basic lithium salts in the lithium industry chain, mainly in the form of anhydrous lithium hydroxide and monohydrate lithium hydroxide.
Battery grade lithium hydroxide monohydrate is mainly used for the preparation of positive electrode materials for lithium-ion batteries.
It can also be used as an additive for alkaline battery electrolytes and in the manufacture of lithium.
Lithium hydroxide, as the core lithium salt downstream of the lithium industry chain, is an important raw material in the field of batteries,
especially the high nickel ternary cathode material widely used in high-performance batteries, which is an indispensable core lithium source in its production.
In the field of batteries, high nickel ternary materials are mainly divided into NMC811 and NCA.
Chinese companies mainly produce NMC811, while Japanese and Korean companies mainly produce NCA.
Specific NMC811 material manufacturers in China, please refer to the top 5. High nickel ternary cathode material company in China.
Currently, many new energy vehicles equipped with high nickel ternary batteries have a battery life of over 500 kilometers.

Production process of lithium hydroxide
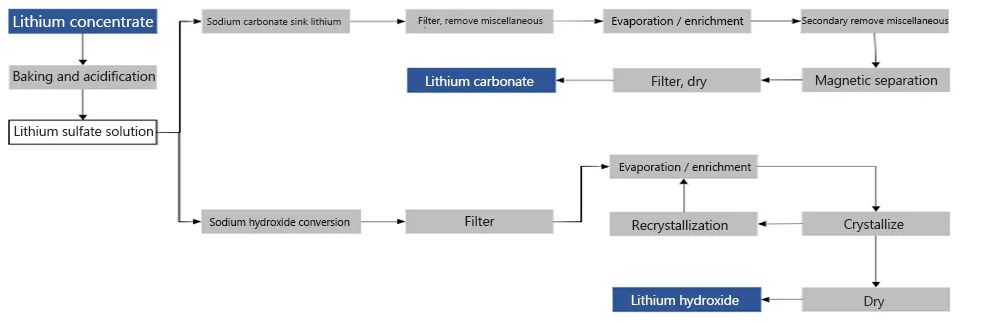
From the perspective of production technology, lithium hydroxide can be produced in one step after acidification of lithium sulfate,
or lithium carbonate can be produced by extracting lithium from salt lake salt water first, and then lithium hydroxide can be produced by catalytic method.
Therefore, considering the cost of producing lithium hydroxide from pyroxene, it is relatively advantageous.
Due to the significant cost advantage of salt lake systems in lithium carbonate, and the quality advantage and no cost disadvantage of ore systems in lithium hydroxide,
the new production capacity of lithium compounds in ore systems has mainly been concentrated on battery grade lithium hydroxide production lines since 2019.
However, the new production capacity of mica lithium extraction is mainly concentrated on the lithium carbonate production line,
and its "quasi grade" products are a supplement to salt lake lithium extraction.
Extracting lithium from ore
The main disadvantage of extracting lithium from ore is that, except for a few high-grade resource points (such as Talison Green Tree Lithium Mine),
the production of lithium carbonate and lithium chloride products using ore is generally in the middle to high position of the global cost curve.
From the perspective of cost safety, extracting lithium from ore is more suitable for the production of high-quality products such as battery grade lithium hydroxide.
Extracting lithium from salt lake saltwater
As far as mature technologies are concerned,
extracting lithium from salt lakes is more suitable as a low-cost raw material basis for producing basic lithium salts
such as industrial grade/battery grade lithium carbonate and lithium chloride.
The production of lithium hydroxide requires carbonization based on lithium carbonate products.
But in the future, if electrolysis, bipolar membrane and other processes are mature, it will help salt lakes achieve one-step direct production of high-quality lithium hydroxide products.
Comparison between lithium carbonate and lithium hydroxide
Lithium carbonate and lithium hydroxide are both raw materials for batteries.
In the market, the prices of lithium carbonate and lithium hydroxide have been consistently rising and falling.
What is the difference between these two materials?
Comparison with the Preparation Process
Both can be extracted from Spodumene and the cost is not significantly different, but if they are converted to each other,
additional costs and equipment are required, and the cost-effectiveness is not high.
The technical routes are different. The preparation of lithium carbonate mainly adopts the sulfuric acid method.
Lithium sulfate is obtained through the reaction of sulfuric acid and spodumene.
Sodium carbonate is added to the lithium sulfate solution and then separated and dried to prepare lithium carbonate;
Lithium hydroxide is mainly prepared by alkaline method, that is, using sodium fluoride and calcium hydroxide.
Some people also use the so-called sodium carbonate pressurization method, which first prepares a lithium containing solution,
then adds lime to the solution, and finally prepares lithium hydroxide.
In short, spodumene can be used to prepare lithium carbonate and lithium hydroxide simultaneously,
but the process routes are different, the equipment cannot be shared, and the cost difference is not significant.
In addition, the cost of preparing lithium hydroxide from salt lake salt water is much higher than that of lithium carbonate.

The technical difficulty of converting lithium carbonate into lithium hydroxide is relatively small, but the cost and construction period are relatively high.
Preparation of lithium hydroxide from lithium carbonate as raw material.
It participates in the reaction of calcium hydroxide in lithium carbonate to produce lithium hydroxide.
This process is quite complex.
However, it is necessary to establish a special production line. The production cost per ton is at least 6000 RMB, without considering depreciation and other factors.
Considering factors such as environmental impact assessment, the construction period should be at least 1-2 years.
When the price of lithium carbonate is higher than that of lithium hydroxide,
the lithium carbonate flotation method directly sells lithium carbonate without further producing lithium hydroxide.
The preparation of lithium carbonate from lithium hydroxide is relatively simple, but it also requires additional costs.
Add carbon dioxide to lithium hydroxide solution to obtain lithium carbonate solution, then separate, precipitate and dry to obtain lithium carbonate solution.
Similarly, this process requires the construction of a dedicated production line, which also incurs additional costs.
Comparative applicability
Due to the lower sintering temperature required for high nickel ternary batteries,
lithium hydroxide has become an essential lithium salt for preparing high nickel ternary materials.
The preparation of lithium iron phosphate (LFP) by hydrothermal method also requires the use of lithium hydroxide.
NCA and NCM811 must use battery grade lithium hydroxide, while NM622 and NCM523 can use lithium hydroxide or lithium carbonate.
Generally speaking, products produced with lithium hydroxide exhibit superior performance. especially:

Sintering temperature: The sintering temperature of series 8 and above ternary cathode materials is usually low.
If lithium carbonate is used as the lithium source, incomplete decomposition may occur due to insufficient sintering temperature,
excessive free lithium on the positive electrode surface,
strong alkalinity, and increased sensitivity to humidity;
Therefore, high nickel ternary cathodes typically use lithium hydroxide as the lithium source.
The use of lithium hydroxide and lower sintering temperatures can also reduce cation mixing and improve cycling stability.
In contrast, the sintering temperature of lithium carbonate usually needs to be above 900 ℃ to obtain stable materials, and it is difficult to use as a lithium source for high nickel materials.
Emission capacity/faucet density: Lithium hydroxide, as a lithium source material, has a first discharge capacity of up to 172Mah/g,
with high faucet density and high charging and discharging performance.
Consistency: Compared with lithium carbonate, lithium hydroxide has the advantages of good stability and consistency,
making it more suitable for high-end cathode materials.
Cycle life: Ternary materials prepared with lithium hydroxide as the lithium source have more uniform particles, greatly improving the cycle life of ternary materials.
Generally speaking, battery grade lithium hydroxide of ternary materials is superior to lithium carbonate.






